Our ApproachTreating the Whole Body
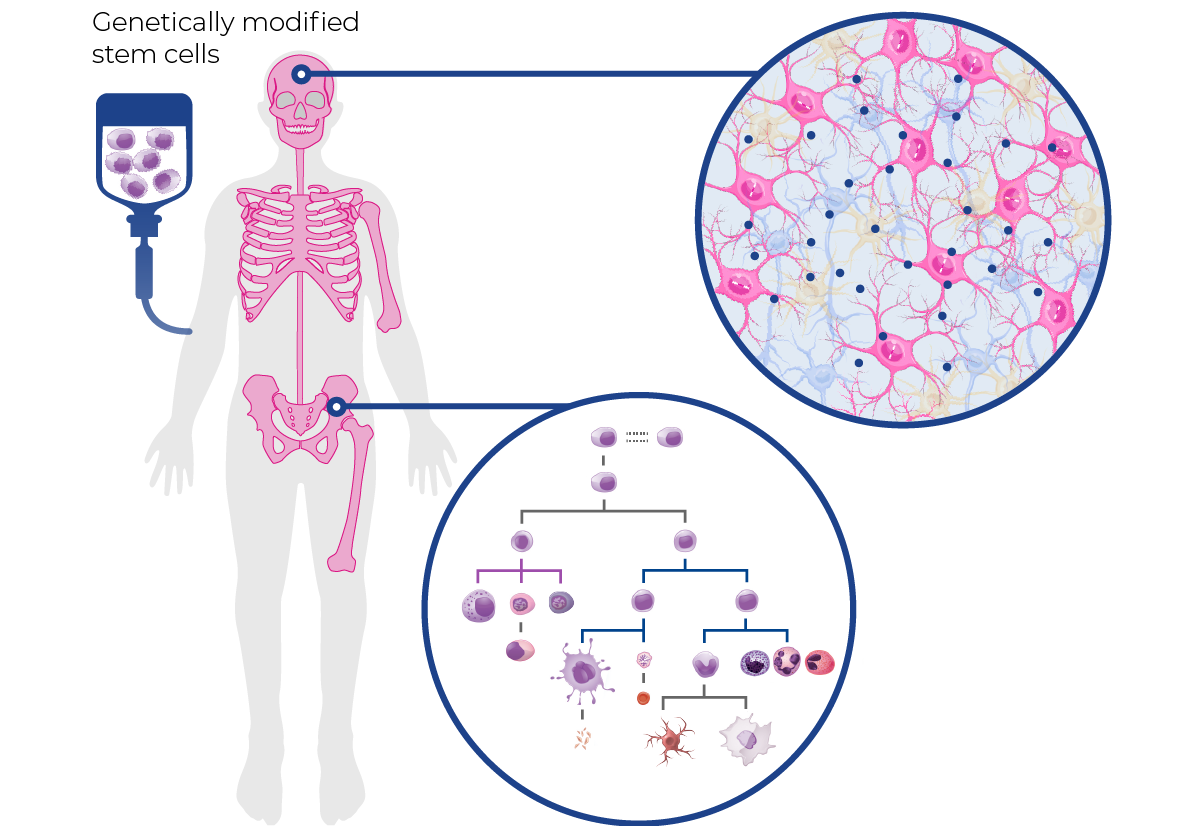
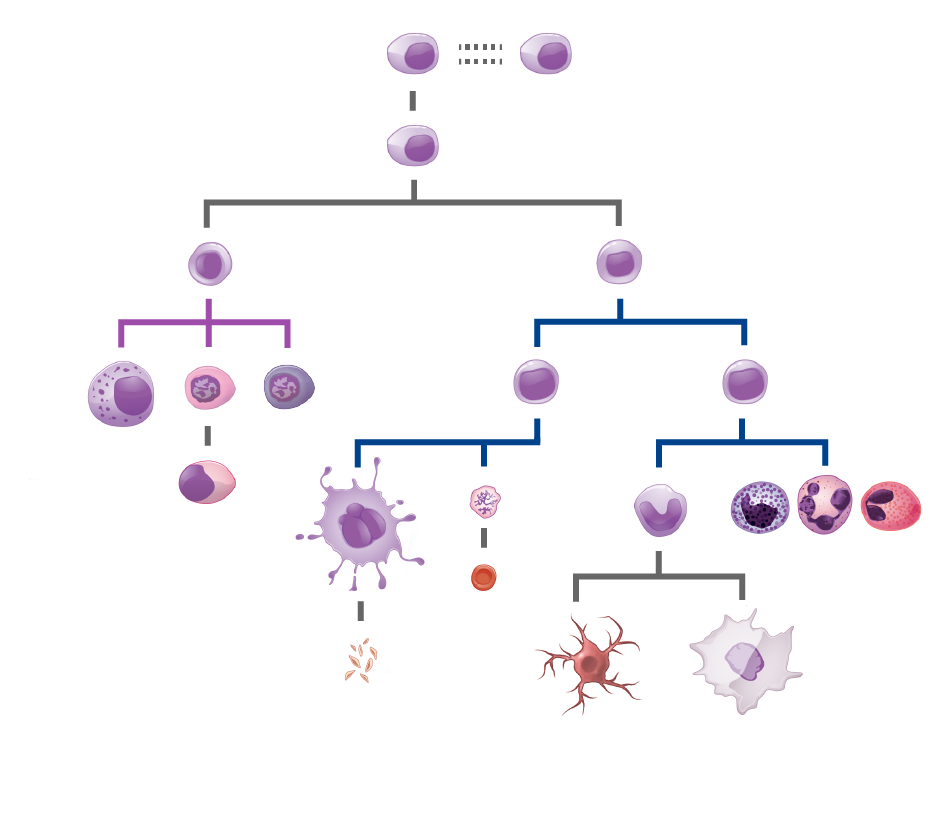
ONE therapeutic gene: distributed head-to-toe
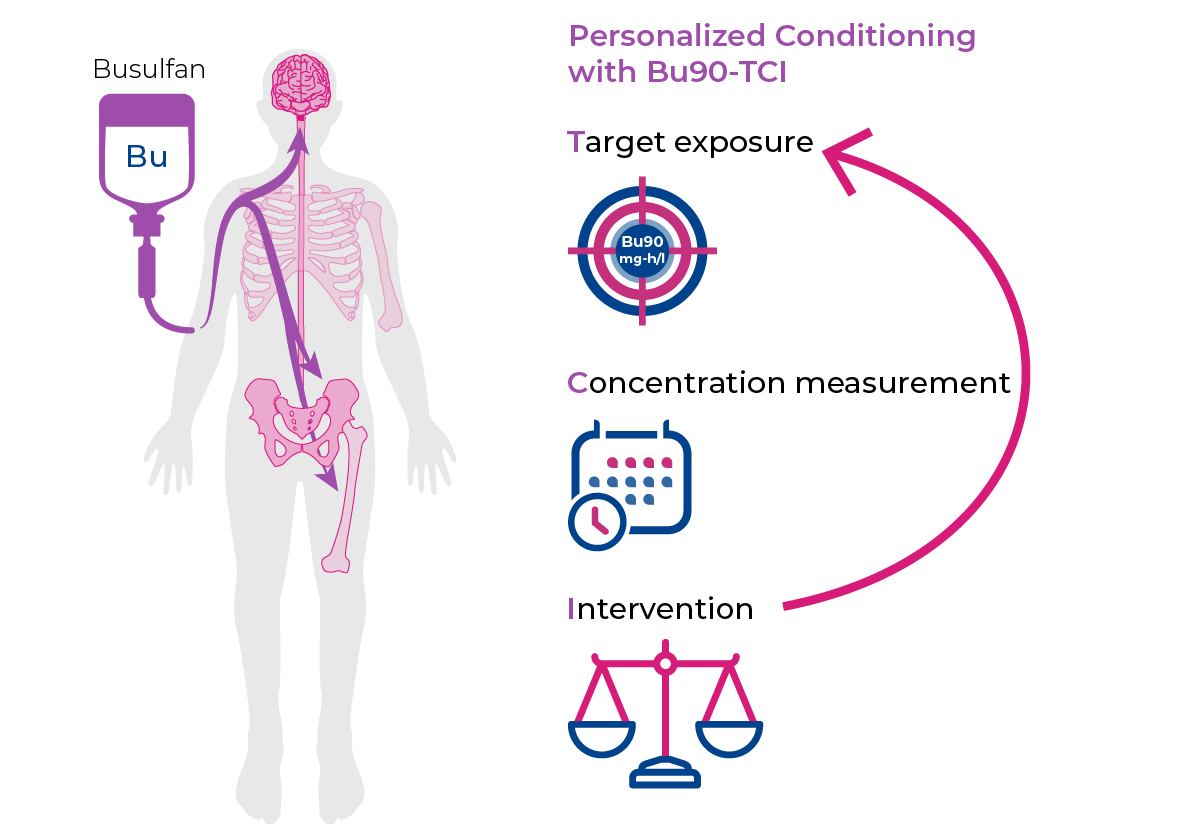
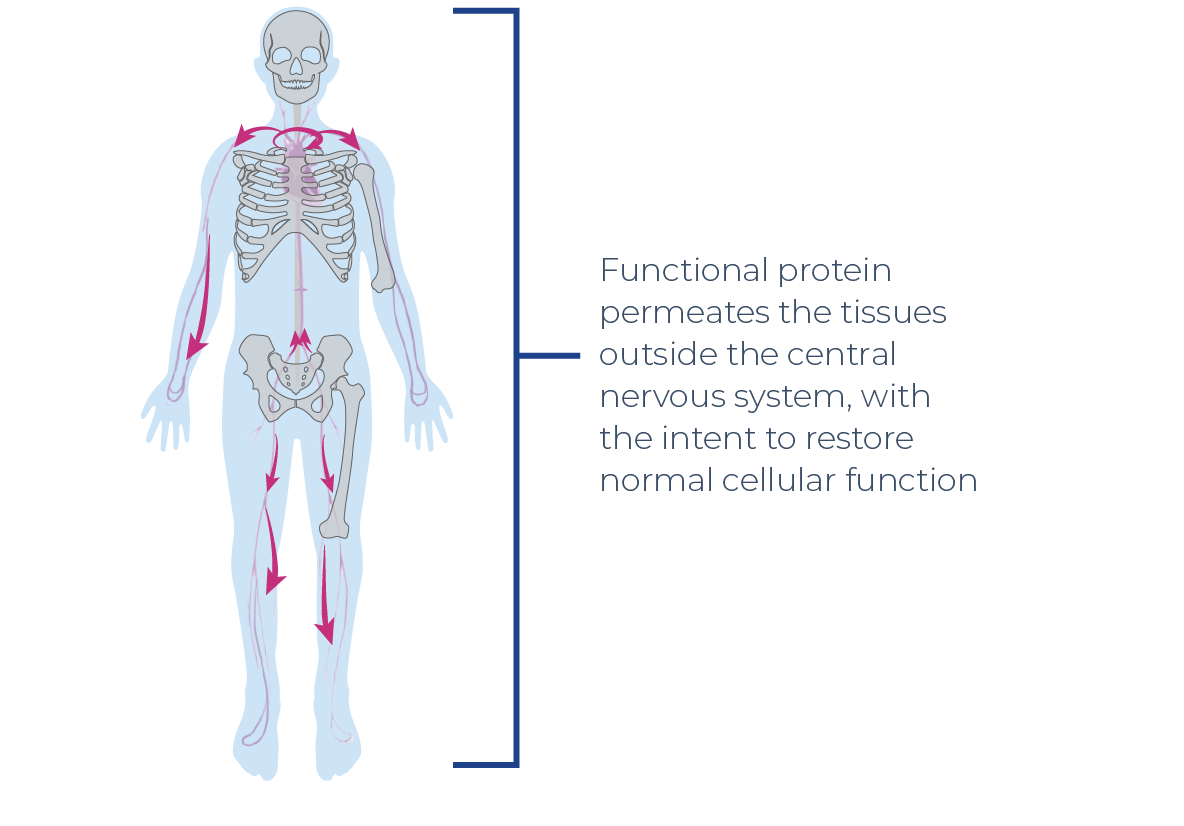
Our investigational gene therapies are being studied to assess their impact on every tissue in the body, including the brain and spinal cord.
Our gene therapies are investigational and have not been approved by FDA or any other regulatory agency. Their safety and efficacy are being studied in clinical trials.