
Moonlighting proteins: A surprising finding in a clinical trial suggests new insights into gene therapy
Science is full of surprises – it’s one of the things I love about this work.
I’ve been reminded of this recently in light of an initial observation from the Phase 1/2 clinical trial[1] of AVR-RD-04, our investigational lentiviral gene therapy for cystinosis. Sponsored by our academic collaborator Stephanie Cherqui, Ph.D. at the University of California, San Diego, the trial has dosed three patients so far.
Early, interim data from the first three patients have generally been in line with our expectations. But we also noticed that one of our trial participants underwent a notable change in appearance, with skin, eyebrows and hair gaining a progressively deeper color as the months went by. It was a pleasant surprise for the young man, who remarked that the rest of his family also has that brunette coloring: he had previously stood out with pale skin and blond hair and eyebrows.
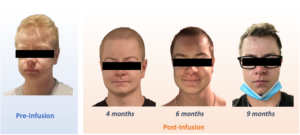
It was a welcome scientific observation for our team, as well. The darker pigmentation in this case was not just skin-deep – it was a potential indication of the multiple roles the protein cystinosin plays – including in pigment production – outside its day job keeping lysosomes cystine-free.
I believe that understanding and embracing the potential of “moonlighting proteins” may be one key to helping advance the field of gene therapy.
The marvel of the multi-tasking protein
First, some background: We humans are incredibly complex organisms, and so are the proteins that do most of the work to keep our cells functioning properly. The term ‘Moonlighting proteins’ refers to proteins that perform a variety of independent and often unrelated functions.[2] Several hundreds of moonlighting proteins with a large variety of functions have been identified and many are suspected to play a central role in numerous diseases.[3]
Cystinosis is a degenerative disease, caused by a single mutation on the CTNS gene, that affects approximately 1 in 100,000 to 1 in 200,000 people.[4] The CTNS gene encodes for the cystinosin protein, which is responsible for transporting the amino acid cystine out of lysosomes. When the gene is mutated, cystine builds up in the lysosomes and this accumulation of cystine forms crystals, which can cause tissue and organ damage.[5]
Cystinosin’s day job is as a transporting protein: it helps recycle organelles within all the cells of the body. When moonlighting, it is involved in the production of pigmentation – as the clinical trial participant I mentioned earlier experienced first-hand.
But what is the potential significance of those changes to skin, eyebrow and hair color?
These observations suggest that cystinosin is functioning as a transporter protein and performing a variety of other unrelated functions, such as in pigment production, just as it would in healthy individuals.
We believe this finding has an implication beyond cystinosis, and beyond investigational AVR-RD-04. Moonlighting proteins are common with enzymes that are involved in metabolism, likely including lysosomal disorders. Keep in mind that the gene therapy approach is designed to replace the whole protein that is missing, and therefore all of the missing protein’s functions, even though we don’t always know what all of the functions are. With drugs, we generally replace just one known function of the protein.
For instance, the protein glyceraldehyde-3-phosphate dehydrogenase (GAPDH) has been shown to be associated with at least 11 functions, including promoting apoptosis or cell death.[6] GAPDH has been associated with Alzheimer’s disease – and may possibly play a role in other neurodegenerative diseases, such as Huntington’s and Parkinson’s diseases. These are highly complex illnesses with diverse manifestations. Perhaps moonlighting proteins like GAPDH are a common thread across the many symptoms. If so, providing functional protein might be the only route to comprehensively treating such disorders.
It’s exciting to think that the biotech field might be able to have a beneficial impact on multiple aspects of a devastating disorder, such as TREM-2 Alzheimer’s disease, by restoring innate production of just one protein.
At AVROBIO, we stress the potentially broad reach of gene therapy and the importance of treating the whole body, from head to toe. The phenomenon of moonlighting proteins reminds us – and our colleagues in the field – that we must look for ways to measure all potential benefits of investigational gene therapies, beyond straightforward biomarkers of disease such as the accumulation of toxic substrate in tissues and organs. Studying those diverse impacts, in turn, would help us learn so much more about the disease – while developing ways we can potentially treat the disease, as well. We at AVROBIO have already departed on the journey…
[1] Collaborator-sponsored Phase 1/2 clinical trial of AVR-RD-04 is funded in part by grants to UCSD from the California Institute for Regenerative Medicine (CIRM), Cystinosis Research Foundation (CRF) and National Institutes of Health (NIH).
[2] Daphne H.E.W. Huberts, Ida J. van der Klei, (2010). Moonlighting proteins: An intriguing mode of multitasking, Biochimica et Biophysica Acta (BBA) – Molecular Cell Research, 1803(4), 520-525, https://doi.org/10.1016/j.bbamcr.2010.01.022.
[3] Jeffery C. J. (2018). Protein moonlighting: what is it, and why is it important?. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 373(1738), 20160523. https://doi.org/10.1098/rstb.2016.0523.
[4] Nesterova, G., & Gahl, W. A. (2001). Cystinosis. In M. P. Adam (Eds.) et. al., GeneReviews®. University of Washington, Seattle.
[5] Wilmer, M. J., Schoeber, J. P., van den Heuvel, L. P., & Levtchenko, E. N. (2011). Cystinosis: practical tools for diagnosis and treatment. Pediatric nephrology (Berlin, Germany), 26(2), 205–215. https://doi.org/10.1007/s00467-010-1627-6.
[6] Sriram G, Martinez JA, McCabe ER, Liao JC, Dipple KM (Jun 2005). “Single-gene disorders: what role could moonlighting enzymes play?”. American Journal of Human Genetics. 76(6): 911–24. doi:10.1086/430799. PMC 1196451. PMID 15877277.