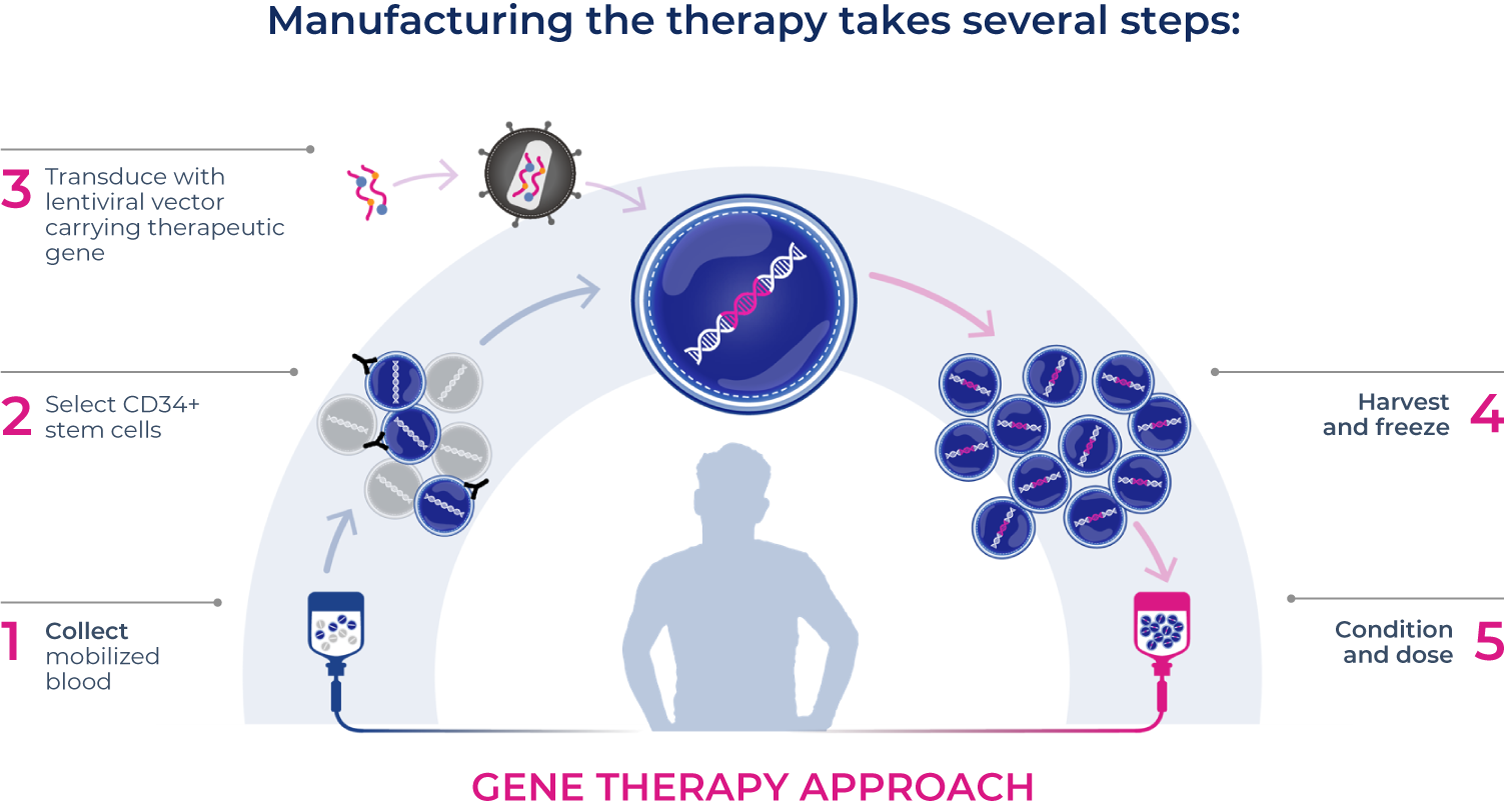
Our
Approach
ONE GENE. One-time therapy.
We strive to halt, reverse or even prevent lysosomal disorders with a single dose.
Our investigational therapies are designed to enable patients' own bodies to begin producing the functional protein they lack. Importantly, they are also designed so the impact of the therapy potentially extends even to hard-to-reach tissues and organs, like the brain, muscle and bone.